|
I think this is when white light is used that you get an Absorption Spectra. All the colors of the Absorption Spectra do make it kind of confusing. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. Remember how we heated the hydrogen to give the atoms energy By doing that, we excited the electrons in the atom. And these are being absorbed (with emphasis on blue). In 1913, a Danish physicist, Niels Bohr (18851962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. The bright lines are called emission lines. Actually, if you just burned hydrogen and looked at its spectra, you would get the Emission Spectra and not the Absorption Spectra, and this Emission Spectra would only show the bunch of blue lines, one purple line, and one red line. All the other colors shown are just part of the natural light being shown down on the element. This is the color that will be the opposite of the flame color on the color wheel. Remember, always look at the color area on the rainbow that is blacked out the most. Why the distances between the lines for the hydrogen spectrum decreases with the decrease in wavelength Just one. So if blue is being absorbed, the opposite color would be transmitted and this color is orange. Atomic emission spectrum is non-contiuous and it is named as line spectrum. However, there are MORE dark lines in the blue region. If you look at the lines for hydrogen blue, purple, and red are being absorbed. Therefore, all the other colors would be absorbed. (This would be orange.) The element hydrogen turns orange when being burned and this color is transmitted to us. This means that if there is a big dark band where blue would be, then the opposite color to blue on the color wheel is being transmitted. You are supposed to look at the dark areas of the absorption spectra and those dark areas indicate that the color which would be there is being absorbed. Assign these wavelengths to transitions in the hydrogen atom. Note how they are complete inverses of each other.I think both the absorption and emission lines are showing which colors are being absorbed. Hydrogen-like atoms are those atoms with only one electron remaining, regardless of the number of protons in the nucleus. An atomic emission spectrum of hydrogen shows three wavelengths: 1875 nm, 1282 nm, and 1093 nm. I am attaching the images of the emission and absorption spectra of hydrogen atom. 
For absorption spectroscopy, varying wavelengths of photons are incident on the atom and the corresponding photon is absorbed by the atom resulting in no photon of that particular wavelength being detected. Sir William Huggins identified 10 lines as being emissions of atomic hydrogen. When atoms releases photons (light), the detector catches the production of the photon and gives out emission spectra. What kind of spectrum you have at hand depends on which instrument and detector you are using. When a photon is absorbed, the electron uses this energy to excite to a higher energy state. 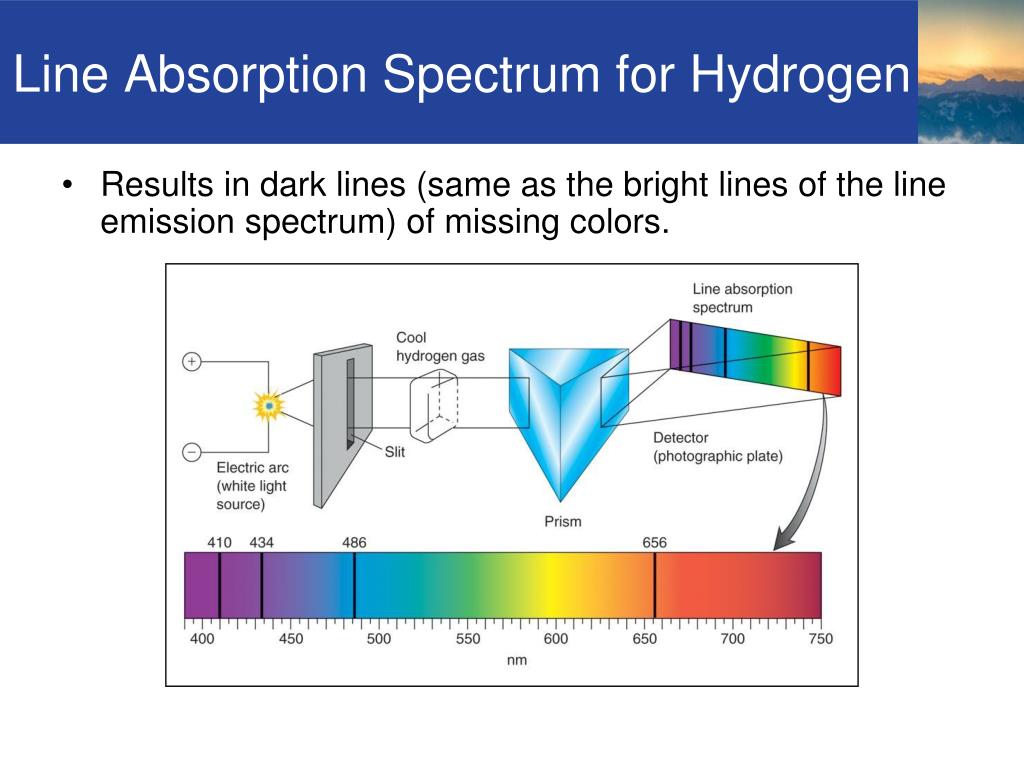
Calculate the energy of each line to four significant figures in Joules using Eq. 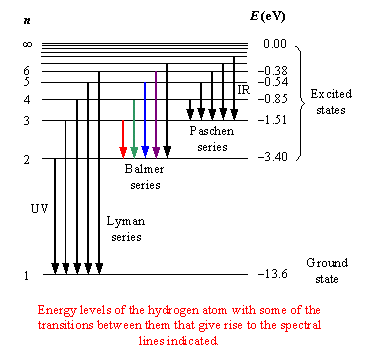
However, the processes by which these two spectra are generated are completely the opposite of each other.Īn atom emits a photon when an electron goes from a higher energy state to a lower energy state, thereby releasing energy in the form of light. The lines of the hydrogen atomic emission spectrum in the visible are known to be at 410.1, 434.0, 486.1, and 656.2 nm. So, naturally, if an atom exhibits some emission spectrum, it will also give an absorption spectrum. Atomic Spectrum Finger Prints of an Atom These are called line emission spectra The light is emitted given off Prepared By: Sidra Javed 7. Emission and absorption are inverse processes. The spectrum consists of separate lines corresponding to different wavelengths.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
